 
Copper has two naturally occurring isotopes. Neutrons and protons, commonly callednucleons, are bound together in the atomic nucleus, where they account for 99.9 percent of the atoms mass. If the average atomic mass of chlorine is 35.46 amu and its natural abundance is 75.53%, what is the atomic mass of the Cl-37 isotope? Their masses are 10.0129 amu and 11.00931 amu. 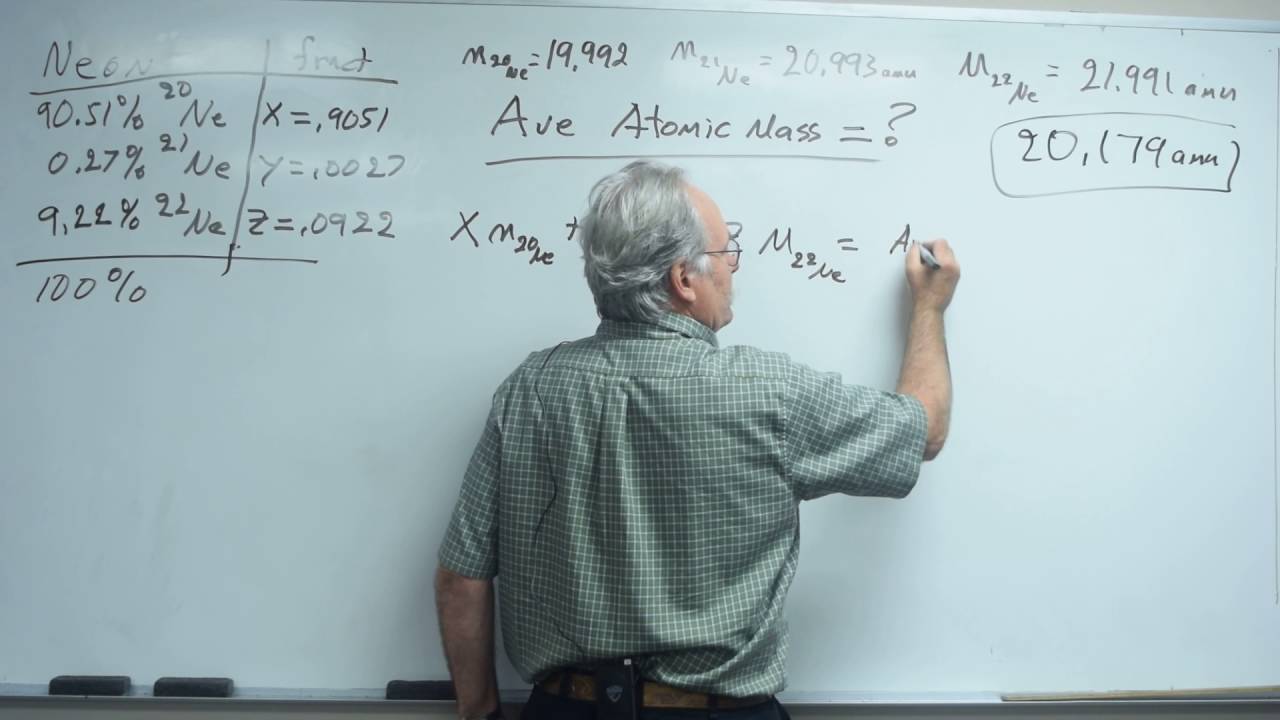
If object A has a mass of 24g and a volume of 2.0mL and object B has a mass of 36g and a volume of 6.0mL. Nitrogen: When reacted with nitrogen, magnesium turns into magnesium nitride. There are 19 radioisotopes that have been discovered, ranging from 18Mg to 40Mg (with the exception of 39Mg). Magnesium has three stable isotopes, Mg-24, Mg-25, Mg-26. A bag of Fritos chips states that 15 chips weigh 28.0 grams and have 10.0 grams of fat. (a) If the natural abundance of Br-79 is 50.69%, what is the natural abundance of Br-81? What are the percent natural abundances of these isotopes? Mg-25 is 10.13%. Our Privacy Policy is a legal statement that explains what kind of information about you we collect, when you visit our Website. In Table PageIndex2>, information about the naturally occurring isotopes of elements with atomic numbers 1 to ten is included. Similarly, thep blockare the right-most six columns of the periodic table, thed blockis the middle 10 columns of the periodic table, while thef blockis the 14-column section that is normally depicted as detached from the main body of the periodic table. What is the relative abundance of the 24Mg isotope? two atoms of different elements must have. Use the isotope abundances and masses to ca. What mass of CaCO3 is required to neutralize 0.0120 mole of HCl according to the following balanced chemical equation? Isotope 2 is found to have an isotopic mass of 40.9618 amu. 1) You may use almost everything for non-commercial and educational use. 
(b) If the natural abundance of Ag-107 is 51.84%, what is the natural abu, Naturally occurring boron, B, consists of two isotopes one is B-10 with isotopic mass 10.012937 u and the other is isotope B-11 with isotopic mass 11.009354 u. Mg 25 and Mg 26 are used to study the absorption and metabolism of magnesium in the human body. There are three stable isotopes of magnesium are 23.9850, 24.9858, and 25.9826 amu. The second isotope has a mass of 70.9247 amu. In a weird parallel universe, the percentages of these isotopes are 45.1%, 13.4%, and 41.5%, respectively. A weighted average you are probably most familiar with in terms of grades, where homework assignments count a certain percent. A sugar solution has a density of 1.30 g/mL. What would be the isotopic symbol for the element argon that contains 22 neutrons? elements that have only five electrons in the highest-energy p orbital are called _. (a) Use the periodic table to find the atomic mass of silver. Identify the number of and type of each atom present in the compound. Naturally occurring isotopes of elemental antimony is a mixture of isotopes. what is the frequency of light that has a wavelength of 511 nm? All everyday objects that can be touched are ultimately composed of atoms, which are made up of interacting subatomic particles, and in everyday as well as scientific usage, "matter" generally includes atoms and anything made up of them, and any particles and objects that act as if they have both rest mass and volume. An element has two naturally occurring isotopes. Magnesium has 3 stable isotopes with masses of 23.98504, 24.98584, and 25.98259 respectively. What is the relative atomic mass of magnesium? Use the periodic table to find the atomic mass of silver. In chemistry and physics, matter is any substance that has mass and takes up space by having volume. The neutron has a mean square radius of about 0.81015 m, or 0.8 fm, and it is a spin- fermion. Because of this, the first two rows of the periodic table are labeled thes block. Rhenium is 62.60% 187Re, and the atomic mass of 187Re is 186.956 amu. One mole of which of the following compounds contains the least number of nitrogen atoms? You can't just add all of your scores together and do a straight average. Most elements have more than one possible oxidation state. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
